Pharma
-

HHS plan to address ‘overprescribing’ of psychiatric medications
While the initiative was greeted with cheers of support at the MAHA Institute summit, many behavioral health specialists warned the effort risks stigmatizing care and reducing treatment options.
-

DOJ signals assertive enforcement of life sciences sector at PCC
For compliance professionals attending the annual conference, the takeaway was hard to miss: 2026 will require a more rigorous compliance framework and transparency.
-

FDA takes steps toward real‑time clinical trials
The regulator said the goal is to speed up the development of promising therapies by addressing longstanding early-phase delays.
-

Rebate debate redux: Revisiting 340B drug pricing
In a high-stakes struggle over the fate of the popular drug pricing program, neither the Trump administration nor safety-net hospital groups seem willing to cede the field.
-
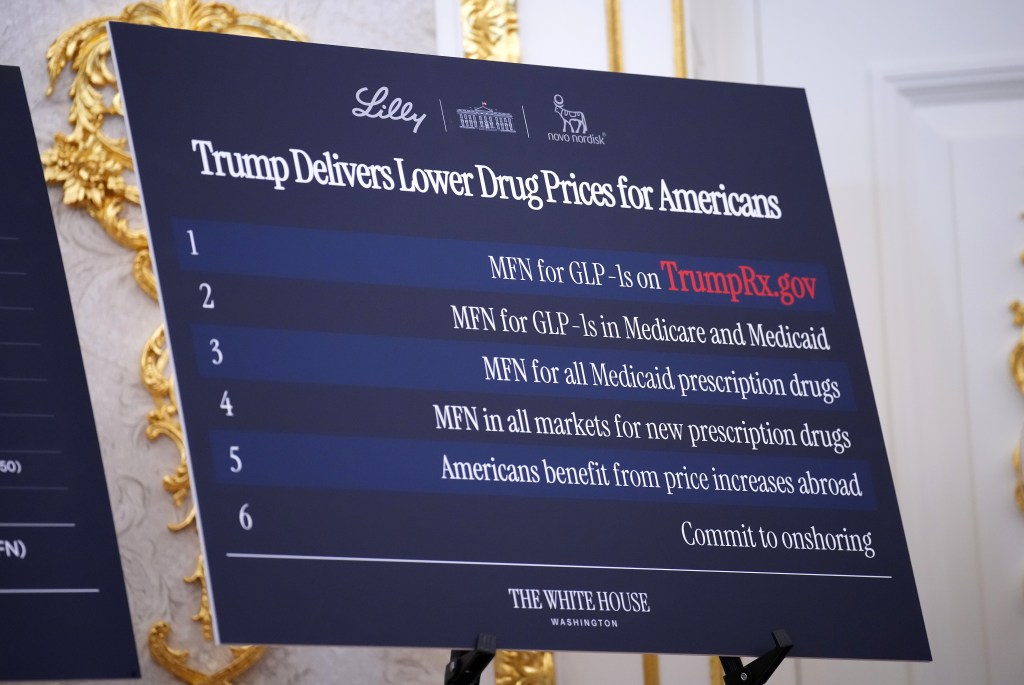
Senate Dems move to force detailed disclosure of Trump’s pharma deals
The legislation would require the Trump administration to reveal the deals it cut with pharma companies to assess whether they deliver real savings or not.
-

White House issues executive order to pave way for psychedelic drug treatments
The order, which surprised some legal analysts, will accelerate access to some Schedule I controlled substances to treat patients with serious mental illness.
-

FDA to consider relaxing restrictions for peptides
The regulator said it is looking to take seven peptides off a restrictive list for unapproved drugs considered too risky.
-

What the ECJ’s landmark ruling on defective products liability means for life sciences manufacturers
This article covers the expanded litigation risk, the steps life science companies should take to reinforce their product safety, and risk‑management governance.
