Pharma
-

Badly fitting risk – lessons from impact investment
When buying off-the-shelf guff policies leads to poor risk management: what to look out for and what to avoid.
-

Pfizer receives PMCPA censure for providing business-class flights to UK HCPs
Ruling reinforces long‑standing Code of Practice expectations on appropriate hospitality, certification, and standards across the pharmaceutical industry.
-

FDA to streamline biosimilar development to lower drug prices
New federal guidance recommends using drugs approved outside of the US as comparators to cut down on pricey pharmacokinetic testing.
-
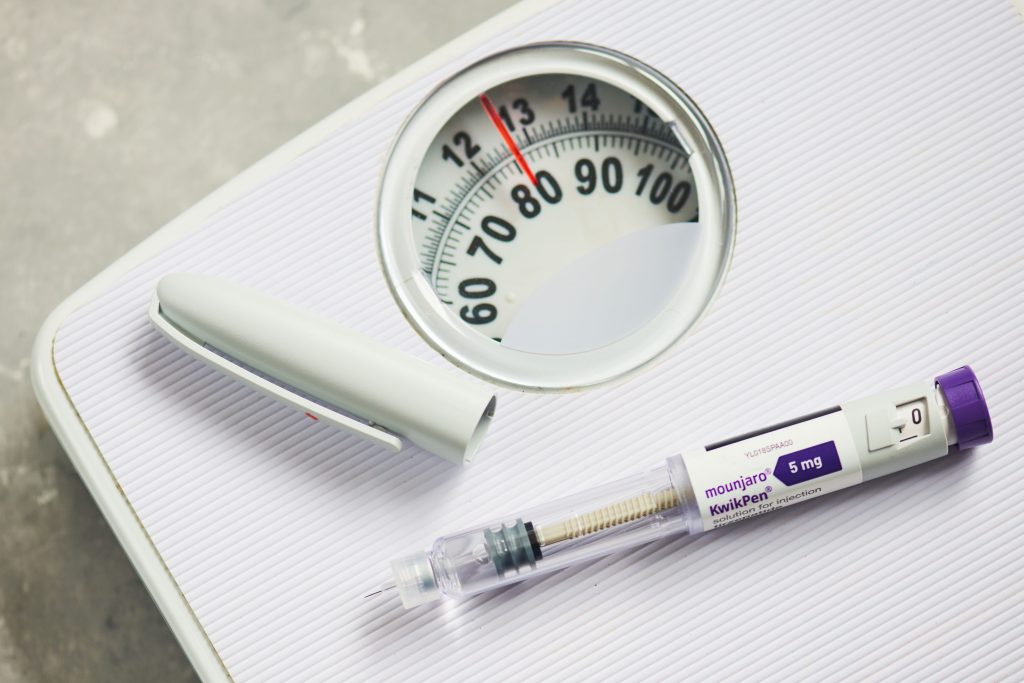
FDA issues warnings to 30 telehealth companies over marketing violations
The letters warning of false or misleading marketing claims point to a transition from the agency’s traditional complaint-driven oversight to a more proactive monitoring system.
-
Changing UK product liability law: potential effects on the healthcare sector
Proactive steps companies in the healthcare sector (or those that insure or invest in those businesses) should take now.
-

Global pharmaceutical syndicate dismantled
The sheer scale of the operation highlights the borderless nature of health-related financial crime.
-

FDA announces new regulatory framework for ultra-rare disease therapies
The guidance will enable the agency to accelerate approval on new treatments for diseases affecting fewer than 1 in 50,000 people.
-
HHS agency signals drug rebate program redux with request for information
The RFI is the Trump administration’s next move to get its drug pricing rebate program back on track after hospital groups blocked it last year.
