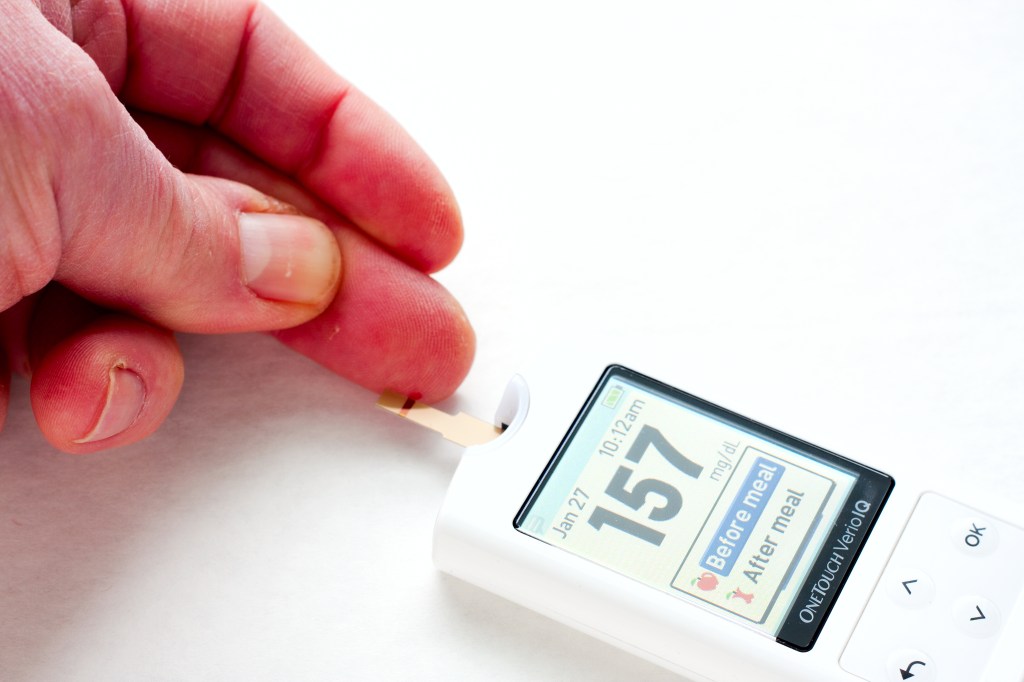
The Centers for Medicare & Medicaid Services (CMS) and the US Food and Drug Administration (FDA) have announced a new regulatory framework designed to accelerate Medicare coverage for certain FDAâdesignated Class II and Class III breakthrough devices.
The goal of the new Regulatory Alignment for Predictable and Immediate Device